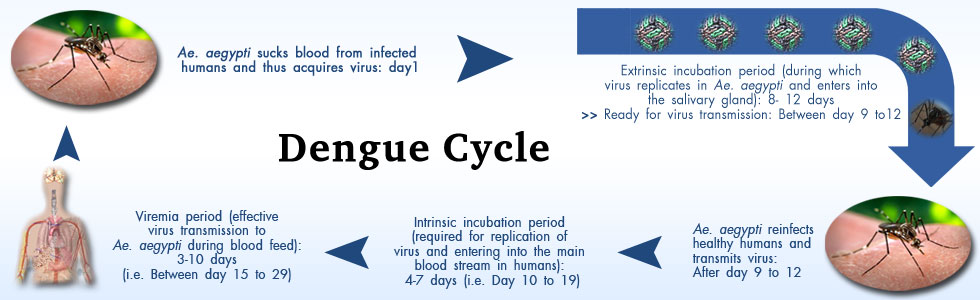
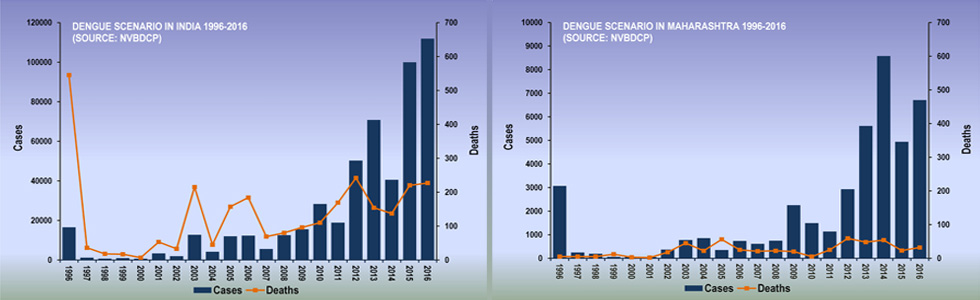
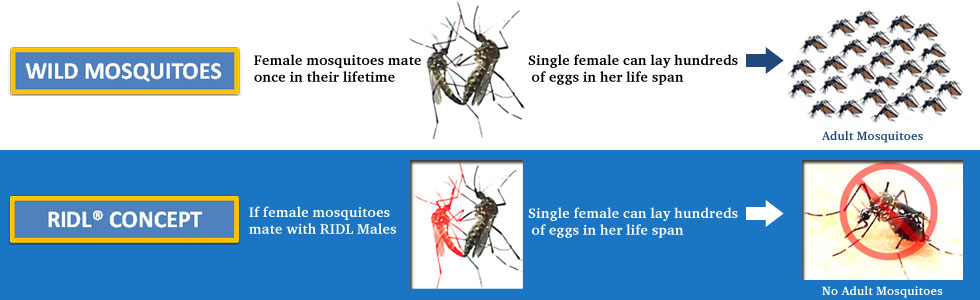
|
Technology GBIT is engaged in R&D activities under the aegis of its RCGM approved Institutional Biosafety Committee (IBSC) to evaluate efficacy of the Self-Limiting and for development of effective phased model for implementation and introduction of the Friendly Aedes aegypti in the integrated dengue mosquito control programme. For this, GBIT has built a state-of-art Arthropod Containment Level II laboratory facility as per the Arthropod containment guidelines to conduct necessary research activities on Aedes aegypti L. mosquitoes. ACL II facility has been approved by the Department of Biotechnology (DBT), Government of India, in 2011. Five efficacy trials have taken place in Brazil, Panama, and the Cayman Islands. These have all demonstrated that releases of Oxitec's self-limiting mosquitoes results in reduction of the wild population by more than 90% – an unparalleled level of control. OX513A has also been successfully evaluated at a number of laboratories including at the Institut Pasteur in Paris and at the WHO Collaborating Centre for Vectors at the Institute for Medical Research (IMR) in Malaysia. OX513A has demonstrated stable performance over 150 generations. |
||